Synthesis of Indole-Oxadiazole coupled isoxazole hybrids as potent EGFR targeting anticancer agents
DOI:
https://doi.org/10.62110/sciencein.cbl.2024.v11.651Keywords:
Cytotoxicity, EGFR inhibitory activity, Indole, Isoxazole, 1,2,4-oxadiazol, hybrid drugs, heterocyclic drugsAbstract
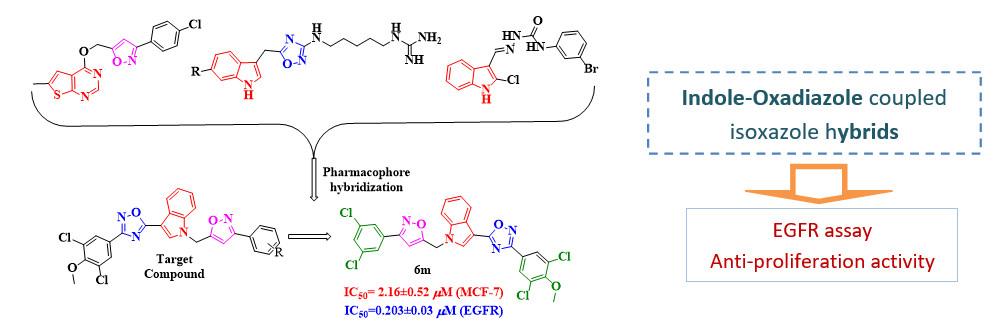
The synthesis of new indole-oxadiazole coupled isoxazole hybrids (6a–o) synthesized by the Cu(I)-catalyzed reaction of in situ generated nitrile oxides with 3-(3,5-dichloro-4-methoxyphenyl)-5-(1-(prop-2-yn-1-yl)-1H-indol-3-yl)-1,2,4-oxadiazole in good yields have been reported here. The chemical structures of all newly synthesized hybrids were confirmed by 1H-NMR, 13C-NMR, and Mass spectra. All synthesized compounds were screened for their in vitro cytotoxicity against two breast cancer cell lines MCF-7 and MDA-MB-231 respectively. All the derivatives were more active against MCF7 than MDA-MB-231 cancer cells and few compounds showed better activity than the standard erlotinib. The ability of more potent compounds to inhibit EGFR tyrosine kinase, one of the key enzymes involved in breast carcinomas was evaluated by in vitro enzymatic assay and it was found that the compound (6g) and (6m) had more inhibitory activity IC50 values 0.311±0.05 and 0.203±0.03 mM than erlotinib (IC50=0.421±0.03 mM).
URN:NBN:sciencein.cbl.2024.v11.651







