3D-QSAR and molecular docking studies of ATP-competitive Akt inhibitors with the scaffold 4-(piperazin-1-yl)pyrimidine
Abstract
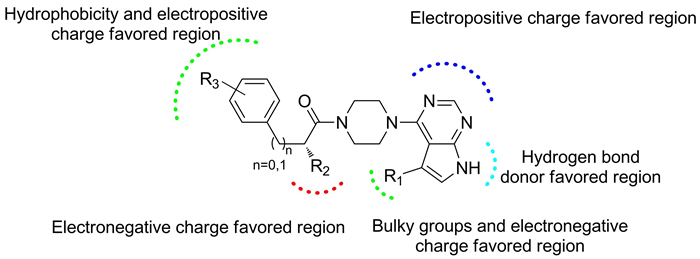
A three-dimensional quantitative structure–activity relationship (3D-QSAR) model of 42 ATP-competitive Akt inhibitors with the scaffold of 4-(piperazin-1-yl)pyrimidine was constructed using the methods of comparative molecular field analysis (CoMFA) and comparative similarity indices analysis (CoMSIA). We finally obtained an optimal CoMFA model with the cross validation value q2= 0.665, the non-cross validation value r2= 0.984 and the predictive r2pred= 0.970. The optimal CoMSIA model built using the combination of electrostatic, hydrophobic and hydrogen bond donor fields yielded the cross validation value q2= 0.718, the non-cross validation value r2= 0.959 and the predictive r2pred= 0.909. Twelve new compounds were designed and their activities were predicted by the constructed CoMFA and CoMSIA models. The result of further docking analysis was in accordance with that of the 3D-QSAR model, validating the predictive reliability of our model. Furthermore, the result of docking also gave insight into the ligand-protein interactions responsible for high inhibitory potency.




